Mitochondrial inheritance diseases paint a complex picture in the vast canvas of medical science. It's a journey where science meets mystery, delving deep into the cellular realms, exploring the unknown, and seeking answers to the myriad of questions surrounding our biological existence.

OPA Heart
The #1 CoQ10 for Heart Health
What causes mitochondrial inheritance diseases?
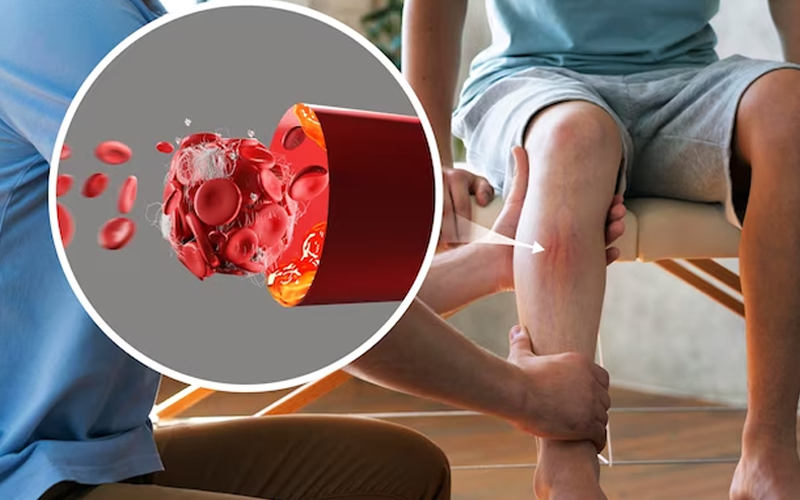
Mitochondrial diseases, mysterious and multifaceted, emerge from the shadows of mitochondrial dysfunction. These cellular powerhouses, the mitochondria, are essential, crafting over 90% of the energy that fuels our bodies and fosters growth. Found in nearly every cell, they are the unsung heroes of cellular function. However, when these vital entities falter, a cascade of cellular misfortune unfolds. Energy production dwindles, cells suffer, and the grim dance of cell death begins. This cellular calamity, when echoed throughout the body, leads to the failure of entire organ systems.
Peeling back the layers, we unearth the root of these disorders in the mutations within mitochondrial DNA (mtDNA) or nuclear DNA impacting mitochondrial function. It's like a once harmonious symphony hitting discordant notes, culminating in a cacophony of symptoms and diseases. The narrative of these diseases is written in the genetic code, with every mutation turning a page to a new chapter of cellular mystery.
Types of Mitochondrial DNA Mutations
The realm of mitochondrial DNA mutations is vast and varied. Among the multitude, large-scale rearrangements stand out, encompassing deletions, duplications, and insertions that alter multiple genes. These rearrangements are like puzzles with missing pieces, leading to a distorted picture of cellular function. On the other hand, point mutations are the subtle whispers of change, affecting a single nucleotide, yet capable of creating ripples in the cellular pond.
Digging deeper, we encounter another type: missense mutations, where a single DNA base change results in a different amino acid, potentially altering protein function. Additionally, frameshift mutations, which insert or delete a base, disrupt the reading frame, leading to widespread changes and often, nonfunctional proteins.
How can I naturally increase ATP levels in my body?

Adenosine triphosphate (ATP) is our body's primary energy source and depleting your cell's supply leads to fatigue. Raising your body's ATP levels may help lessen beneficial effects of this discomfort and ease any feelings of worn outness or discontentment.
Coenzyme Q10, for instance, has been widely shown to improve mitochondrial function and increase ATP levels. Coenzyme Q10 acts as a cofactor in both Krebs cycle and electron transport chain, helping create ATP from metabolic waste products such as fat. Unfortunately, its levels naturally decrease with age but supplementation can restore them.
Acetyl-L-Carnitine has proven its effectiveness by improving cognitive function and alleviating symptoms associated with fibromyalgia. Succinic acid, more commonly known as succinate, increases ATP production through increasing glucose utilization by mitochondria as well as oxygen consumption.
Creatine is an invaluable ATP booster for athletes as it has significant increases in the rate of production and replenishment during intense workouts. D-Ribose supplements, on the other hand, support creation very rapidly - thus being used by hardcore bodybuilders to ease muscle fatigue and speed recovery time.
Causes and Consequences of Mitochondrial DNA Mutations
Mitochondrial DNA mutations bear tales of both inherited and sporadic origins. Like stories passed down through generations, some mutations are inherited, encoded in the genetic tales we receive from our ancestors. Others are sporadic, emerging spontaneously like unexpected plot twists, adding layers of complexity to the cellular narrative.
The consequences of these mutations are as varied as their origins. They paint a diverse landscape of diseases, each characterized by a unique spectrum of clinical symptoms. From neuromuscular disorders to metabolic dysfunctions, the reach of these mutations is extensive, touching multiple organ systems and weaving intricate patterns of disease.
Mitochondrial DNA Mutations
Venturing deeper into the enigma of mitochondrial DNA mutations, we explore a diverse landscape shaped by inherited and spontaneous mutations. It's a world where genetic alterations bloom into a plethora of mitochondrial diseases, each donning different forms and targeting various organ systems. These mutations are not mere anomalies in the genetic fabric; they are architects of cellular chaos, planting seeds of disorder and dysfunction.
Beyond the visible spectrum of disorders, these mutations act as silent puppeteers, orchestrating a symphony of symptoms and giving rise to a myriad of mitochondrial diseases. The repertoire of these diseases is vast, ranging from energy metabolism disorders to neurodegenerative diseases, each narrating a unique tale of cellular misfortune.
Types of Mitochondrial DNA Mutations
Within the genetic tapestry, mitochondrial DNA mutations manifest in myriad forms, including large-scale rearrangements and point mutations. These genetic alterations, each distinct, impact cellular function in varying degrees, shaping the landscape of mitochondrial disorders.
Exploring further, we encounter tRNA mutations, pivotal players in protein synthesis. These mutations can disrupt the delicate balance of cellular function, leading to a cascade of metabolic dysfunction. Similarly, rRNA mutations affect ribosomal function, impairing the cellular machinery responsible for protein synthesis, thereby contributing to the pathogenesis of mitochondrial disorders.
Causes and Consequences of Mitochondrial DNA Mutations
The dance between the causes and consequences of mitochondrial DNA mutations is intricate and multifaceted. The origins of these mutations, whether inherited or sporadic, are embedded in the genetic code, giving rise to a spectrum of mitochondrial diseases, each with its unique clinical presentation and challenges.
The ripple effect of these mutations is profound, affecting cellular energy metabolism, organ function, and overall health. The diversity of clinical manifestations is extensive, ranging from muscle weakness and neurological impairment to metabolic imbalances and organ failure. Navigating through the myriad of symptoms and understanding the underlying genetic alterations is pivotal in deciphering the complexity of mitochondrial disorders and developing targeted therapeutic strategies.
Mitochondrial Mutations That Contribute to Common Disease Phenotypes
What is the commonest mitochondrial disease? Mitochondrial mutations, often hidden behind the curtain of cellular complexity, play a crucial role in contributing to common disease phenotypes. They are like unseen puppeteers, subtly pulling the strings, shaping various forms of disorders, and influencing the narrative of several prevalent diseases. These mutations weave intricate tales of cellular dysfunction, each thread unraveling a different story of common disease manifestations.
Mitochondrial Mutations in Neurodegenerative Diseases
Mitochondrial mutations have been fingered as culprits in a host of neurodegenerative diseases. They compose a sinister symphony, orchestrating a cascade of cellular dysfunctions, casting shadows on the pathways of conditions like Parkinson’s and Alzheimer’s. In these diseases, the mutations act like silent whispers, subtly disrupting the normal functioning of neurons, leading to a gradual decline in cognitive and motor abilities.
Furthermore, these mutations can affect the energy production in neurons, resulting in increased oxidative stress and neuronal death. The landscape of neurodegenerative diseases painted by mitochondrial mutations is vast and varied, with each mutation contributing to a unique set of symptoms and progression patterns.
Mitochondrial Mutations and Metabolic Disorders
Mitochondrial mutations extend their dance to the realm of metabolic disorders. These mutations waltz through metabolic pathways, altering the rhythmic flow of cellular processes, and contributing to the development of disorders such as diabetes. In metabolic disorders, the mutations act like discordant notes in a melody, disrupting the harmony of energy production and utilization.
The influence of mitochondrial mutations in metabolic disorders is multifaceted. They can lead to insulin resistance, impaired glucose metabolism, and can contribute to the onset and progression of Type 2 Diabetes. The ripple effect of these mutations is profound, influencing various metabolic processes and painting a complex picture of metabolic dysfunction.
Mitochondrial vs. Nuclear DNA

Mitochondrial and nuclear DNA form the dual pillars of cellular genetics, acting as the yin and yang of the genetic world. Existing in a delicate equilibrium, they both play vital, distinct roles, each contributing to the symphony of cellular function. Their collaboration and interaction are like a well-choreographed dance, essential for maintaining cellular health, energy balance, and the overall vitality of an organism.
Differences Between Mitochondrial and Nuclear DNA
The differences between mitochondrial and nuclear DNA are pronounced and pivotal. Mitochondrial DNA, small and circular, is a compact powerhouse, carrying essential information for cellular energy production. In contrast, nuclear DNA, residing within the nucleus, is linear and forms the chromosomes, carrying the bulk of genetic information that dictates the characteristics and functioning of an organism.
Moreover, mitochondrial DNA is maternally inherited, passed down from mother to offspring, while nuclear DNA is a blend of both maternal and paternal inheritance, contributing to the genetic diversity of the offspring. These differences in structure, quantity, and inheritance highlight the unique roles and significance of mitochondrial and nuclear DNA in cellular function and genetic inheritance.
Collaboration and Interaction of Mitochondrial and Nuclear DNA

The collaboration between mitochondrial and nuclear DNA is a genetic ballet, harmonious and essential for life. This interaction is crucial for cellular energy production, as nuclear DNA encodes for many proteins that are integral to the mitochondria’s function. The intricate dance between them ensures the smooth functioning of cellular processes and the well-being of the organism.
Furthermore, any missteps in this dance can lead to disruptions in cellular harmony. Mutations in either form of DNA can affect the other, leading to a cascade of cellular dysfunctions and contributing to the development of a myriad of diseases. The understanding of this interaction is vital for unraveling the mysteries of mitochondrial inheritance diseases and offers a path for potential therapeutic interventions.
What are the inheritance patterns in mitochondrial myopathies?
Mitochondrial inheritance diseases are fascinating examples of the unique genetic tapestry that makes up human life, predominantly showcasing a maternal pattern of inheritance. The complexity of this genetic dance is woven through generations, creating a labyrinthine pattern that is both enigmatic and revealing. As each thread of mitochondrial inheritance intertwines, a story of maternal lineage, genetic risk, and potential disease unfolds. It is within this intricate tapestry that the secrets of mitochondrial myopathies lie, demanding a nuanced understanding for effective genetic counseling and risk assessment.
Maternal Inheritance of Mitochondrial Myopathies
Mitochondrial myopathies predominantly trace their roots through the maternal lineage, setting a distinct rhythm in the genetic symphony. This mode of inheritance is unique, forming a critical chapter in the narrative of these disorders. The mitochondrial DNA, passed exclusively from mother to offspring, becomes the sole narrator of this tale, encoding the information that could potentially lead to the manifestation of various myopathies.
Exploring deeper, it becomes evident that this mode of inheritance harbors significant implications. It means that if a mother carries a mutation in her mitochondrial DNA, all her offspring are at risk of inheriting the disorder, regardless of their gender. However, if the father carries the mutation, the risk becomes null, as the father’s mitochondrial DNA does not contribute to the genetic makeup of the offspring. This intricate dance of inheritance not only shapes the risk landscape for individuals but also crafts the familial patterns observed in mitochondrial myopathies.
Genetic Counseling and Mitochondrial Myopathies
Navigating through the world of mitochondrial myopathies is akin to traversing a complex genetic labyrinth. Genetic counseling for these disorders becomes a beacon of light, guiding families through the intricate pathways of mitochondrial inheritance. It is here that knowledge becomes power, illuminating the paths and helping individuals make informed decisions about their health and future.
Genetic counselors act as the cartographers of this labyrinth, mapping out the genetic terrain, identifying risks, and providing insights into the management of these disorders. They unravel the threads of maternal inheritance, interpret the implications of mutations, and offer guidance on potential interventions and lifestyle adjustments. Through this process, individuals gain a clearer understanding of their genetic heritage, the likelihood of disease manifestation, and the available avenues for management and support.
Genetic counseling also plays a pivotal role in family planning, providing prospective parents with insights into the potential risks and helping them make informed choices. It’s a journey of discovery, unraveling the mysteries of mitochondrial inheritance, and laying the foundation for a proactive approach to health and well-being.
Summary
In the intricate and ever-evolving landscape of genetics, mitochondrial inheritance diseases stand as unique enigmas, offering both challenges and opportunities for medical science. These diseases push the boundaries of our understanding, prompting a continuous quest for knowledge and insight. As we delve deeper into the genetic tapestry, unraveling the threads of inheritance and exploring the dance of mitochondrial and nuclear DNA, we edge closer to unlocking the secrets of these disorders.
The journey to comprehend mitochondrial inheritance diseases is a testament to human curiosity and resilience. It’s a pursuit that intertwines science, genetics, and medicine, striving to illuminate the shadows and bring clarity to the complexities of inheritance. Each discovery, every insight gained, brings us a step closer to understanding the myriad of diseases associated with mitochondrial mutations and opens new doors for therapeutic interventions and management strategies.
In this odyssey of understanding, the unique patterns of inheritance become the compass, guiding us through the genetic maze and shaping the narrative of mitochondrial myopathies. It’s a journey of exploration and revelation, where the dance of DNA becomes the rhythm of life, and the threads of inheritance weave the tapestry of our genetic heritage. As we continue to explore, to question, and to seek, the mysteries of mitochondrial inheritance diseases gradually unravel, offering glimpses into the intricacies of human life and the endless possibilities of medical science.
Frequently Asked Questions
Leigh syndrome is widely recognized as the most common inherited mitochondrial disorder. It's characterized by progressive neurodegeneration and can affect multiple organ systems, leading to a variety of symptoms and complications.
Yes, mitochondrial diseases can indeed be inherited, primarily through maternal inheritance. This unique inheritance pattern sees the mitochondrial DNA passed from mother to offspring, with the father’s mitochondrial DNA typically not being inherited.
While Amyotrophic Lateral Sclerosis (ALS) is primarily a neurodegenerative disorder, research has indicated that mitochondrial dysfunction can play a significant role in the disease's progression, thus drawing a connection between ALS and mitochondrial anomalies.
Alzheimer's disease is classified as a neurodegenerative disorder. However, there's growing evidence to suggest that mitochondrial dysfunction is implicated in the disease’s pathology, contributing to the neurodegenerative process.
Mitochondrial diseases, due to their unique inheritance pattern, do not follow the typical dominant or recessive inheritance seen with nuclear DNA. They are primarily inherited through the maternal lineage, with all children of an affected mother having a risk of inheriting the disorder.